Changes in States of Matter
P.CM.06.11 - Describe and illustrate changes in state, in terms of the arrangement and relative motion of the atoms or molecules.
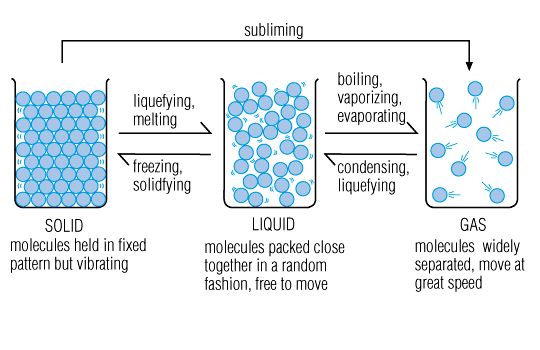
- Most matter on Earth can exist in three states-solid, liquid, and gas.
Liquid: Matter with no definite shape, but a definite volume. The particles in a liquid are not fixed, and can move about.
Gas: Matter that has no definite volume or shape. The particles can move about freely.
Change of State: The physical change from one state of matter to another. Matter can change from one state to another when thermal energy is released.
- Melting: The change of state to a solid to a liquid. At a substances melting point, the particles of a solid are vibrating so fast that they break free of their fixed position.
- Freezing: the change of state from a liquid to solid. At a substances freezing point, the particles of a liquid are moving so slowly that the particles freeze into place.
- Evaporation: If vaporization takes place AT THE SURFACE OF A LIQUID.
- Boiling: VAPORIZATION OCCURS UNDER THE LIQUID
- Condensation: A change from a gas state to a liquid state.
- As the thermal energy of matter increases, its particles spread out and the substance expands.
- Thermal expansion: The expanding of matter when it is heated.